Sutton family's relief as five-year-old's life-saving treatment is approved


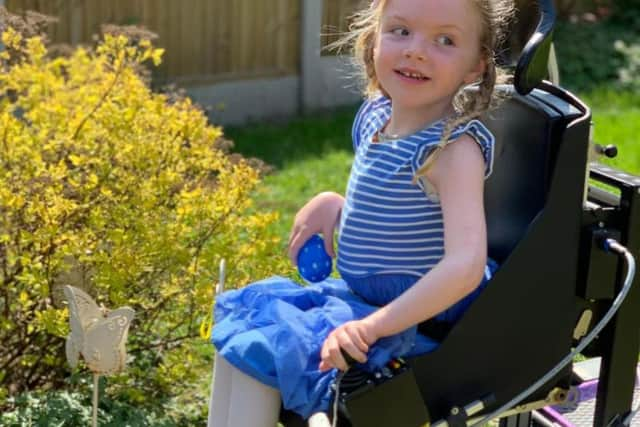
Elsie Novell has spinal muscular atrophy (SMA), which means she is unable to stand or walk. There is currently no cure for Elsie’s condition, but a new drug called Spinraza could provide hope as it halts the symptoms of the disease.
SMA affects the nerves in the spinal cord controlling movement, causing muscle weakness, progressive loss of movement, and difficulty breathing and swallowing.
Advertisement
Hide AdAdvertisement
Hide AdThe family had previously been told by NHS bosses that the vital Spinraza drug would not be made available due to concerns over its long-term effectiveness and its high cost, leading to numerous fundraising campaigns from Charlotte, Elsie's mum, to help the toddler in attending drugs trials as far away as Belgium.


However now, after more than two years of uncertainty, the family have been told that the Spinraza drug is to be made available on the NHS within the coming months - meaning Elsie's life could change forever.
Charlotte, Elsie's mum, says it means her daughter will "no longer start to degrade", and that, even though she is wheelchair-bound, she will start to live a "normal life".
She said: "Since we were told it was not going to be available on the NHS it has been such a rollercoaster of emotions.
Advertisement
Hide AdAdvertisement
Hide Ad"When you get told in diagnosis that your child does not have long, you start to come to terms with it, but when you get hope that a drug could be available it really lifts you up.

"When we found out it was available in America we thought there might be hope, but then getting told it wasn't available here was like diagnosis all over again.
"I woke up yesterday morning (May 15) to an email telling me that it had been approved and my it took a second to really register it.
"This will be amazing for children like Elsie because it means her body can build immunity to illnesses and that she won't live through constant deterioration - it's a massive weight off the shoulders.
Advertisement
Hide AdAdvertisement
Hide Ad"The treatment is done through a lumber puncture which she will need to have forever, but now we won't live in constant fear that she's going to die or get worse.


"Unfortunately it's not going to be a miracle drug and it won't make her muscles start working after five years, but it will trick the body into thinking it can fight disease on its own."
There are currently no active treatments targeting the underlying cause of SMA so the condition is managed through supportive care, which aims to minimise the impact of disability, address complications and improve quality of life.
The treatment will be made available to the youngest and most severely affected (SMA type 1) patients immediately by Biogen, with NHS England offering funding on NICE’s publication of final guidance next month.
Advertisement
Hide AdAdvertisement
Hide AdFor older babies, children and young adults with less severe symptoms (SMA types 2 and 3), the NHS will begin to roll out nusinursen shortly after NICE’s guidance is published.
Meindert Boysen, director of the centre for health technology evaluation at NICE, said: “We are very pleased that we can now recommend Spinraza for people with SMA.
“The committee has recognised that Spinraza is a promising treatment that has been shown to improve a range of outcomes important to patients. But it also recognised that there are significant uncertainties, particularly around its long-term benefits.
“All along we have felt it important to give all parties every opportunity to try to find a way to mitigate these uncertainties in order to make Spinraza available to patients in England."
Advertisement
Hide AdAdvertisement
Hide AdAfter news was revealed about the treatment, Charlotte said the family started to plan a holiday to "take Elsie somewhere she has always wanted to go" - Butlins.
She said: "Elsie has been desperate to go to Butlins for years, so we have organised to take her for the weekend at the end of June in celebration - and we are keeping it a surprise.
"We are also all going on a family holiday to Italy for a wedding later in the summer, so we can all celebrate together."